The other nuclear particle is the neutron. This means that in any atom, the number of protons in the nucleus (often referred to as the nuclear charge) is balanced by the same number of electrons outside the nucleus. Protons are the carriers of positive electric charge in the nucleus the proton charge is exactly the same as the electron charge, but of opposite sign. Atomic Structure All substances are made of tiny particles of matter called atoms Each atom is made of subatomic particles called protons The protons and. For example, any atom that contains six protons is the element carbon and has the atomic number 6, regardless of how many neutrons or electrons it may have. The atomic models of the complex between rabbit skeletal muscle actin and bovine pancreatic deoxyribonuclease I both in the ATP and ADP forms have been. The positive charges equal the negative charges, so the atom has no overall charge it is electrically neutral. This is the defining trait of an element: Its value determines the identity of the atom. An atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The nucleus is itself composed of two kinds of particles. Table 2.2 The number of protons in the nucleus of an atom is its atomic number (Z). If the atoms of a material are ordered in a. 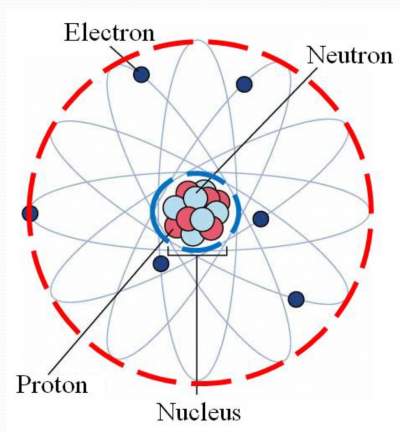
An atom comprises three different particles, known as protons, neutrons, and electrons. \): The structure of the nuclear atom with a central nucleus and surrounding electrons. The atomic or molecular structure of solid materials can generally be found in two states: ordered and disordered. Simply put, atomic structure refers to the structure of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
